Family of Metals With the Second Highest Reactivity
Chapter eighteen. Representative Metals, Metalloids, and Nonmetals
18.1 Periodicity
Learning Objectives
Past the end of this section, you lot will be able to:
- Classify elements
- Make predictions about the periodicity backdrop of the representative elements
We begin this section by examining the behaviors of representative metals in relation to their positions in the periodic table. The chief focus of this department will be the awarding of periodicity to the representative metals.
It is possible to split up elements into groups according to their electron configurations. The representative elements are elements where the s and p orbitals are filling. The transition elements are elements where the d orbitals (groups 3–eleven on the periodic table) are filling, and the inner transition metals are the elements where the f orbitals are filling. The d orbitals fill with the elements in grouping 11; therefore, the elements in grouping 12 qualify as representative elements because the last electron enters an due south orbital. Metals among the representative elements are the representative metals. Metallic graphic symbol results from an element's ability to lose its outer valence electrons and results in high thermal and electric electrical conductivity, amongst other physical and chemical properties. At that place are twenty nonradioactive representative metals in groups ane, two, 3, 12, 13, 14, and 15 of the periodic tabular array (the elements shaded in yellow in Figure one). The radioactive elements copernicium, flerovium, polonium, and livermorium are also metals but are across the scope of this affiliate.
In addition to the representative metals, some of the representative elements are metalloids. A metalloid is an element that has properties that are betwixt those of metals and nonmetals; these elements are typically semiconductors.
The remaining representative elements are nonmetals. Dissimilar metals, which typically form cations and ionic compounds (containing ionic bonds), nonmetals tend to form anions or molecular compounds. In general, the combination of a metal and a nonmetal produces a table salt. A salt is an ionic compound consisting of cations and anions.
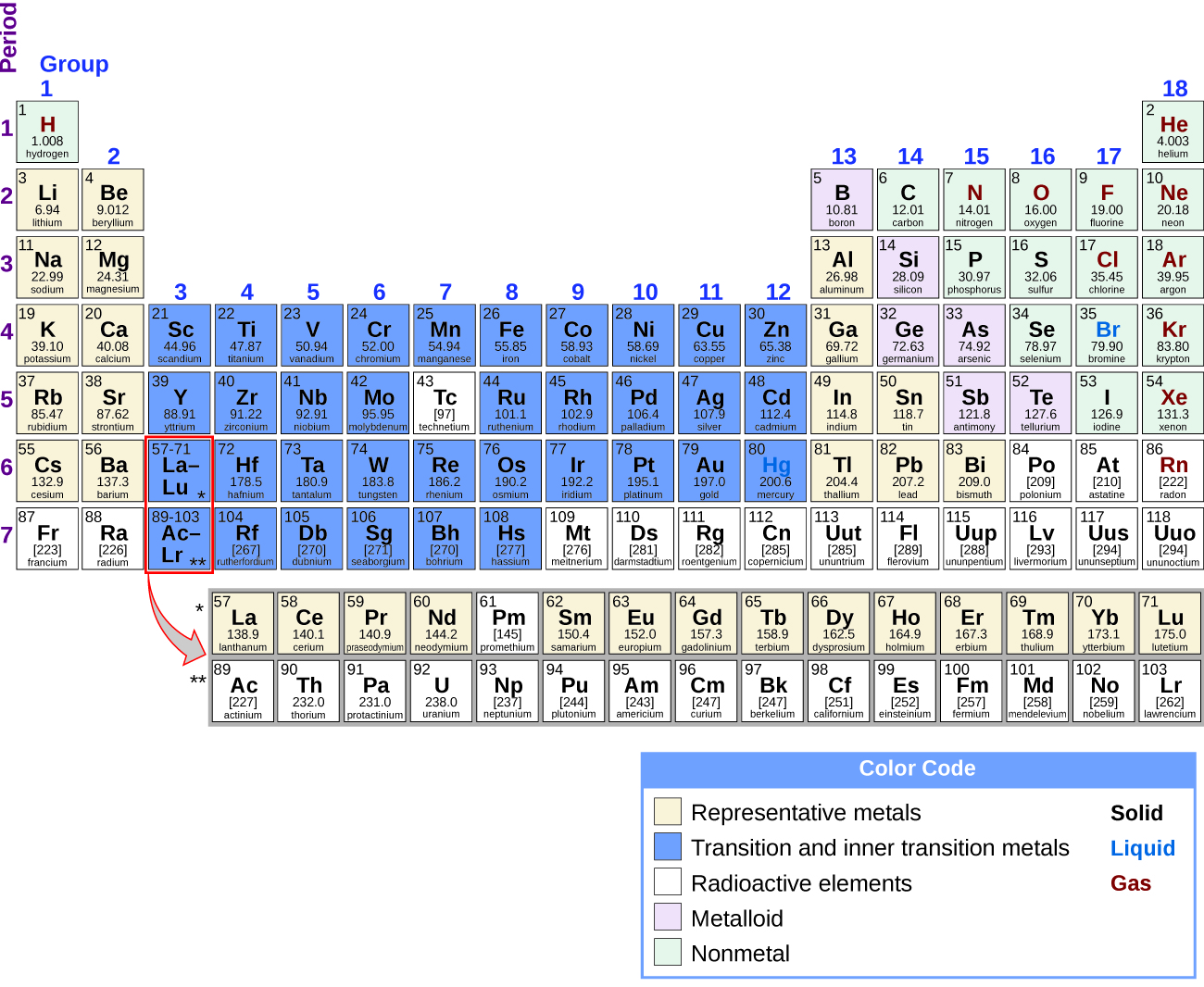
Most of the representative metals do not occur naturally in an uncombined country because they readily react with water and oxygen in the air. Even so, it is possible to isolate elemental beryllium, magnesium, zinc, cadmium, mercury, aluminum, tin can, and lead from their naturally occurring minerals and use them because they react very slowly with air. Part of the reason why these elements react slowly is that these elements react with air to form a protective blanket. The germination of this protective coating is passivation. The coating is a nonreactive film of oxide or another chemical compound. Elemental magnesium, aluminum, zinc, and tin are important in the fabrication of many familiar items, including wire, cookware, foil, and many household and personal objects. Although glucinium, cadmium, mercury, and atomic number 82 are readily available, there are limitations in their use considering of their toxicity.
Group one: The Alkali Metals
The alkali metals lithium, sodium, potassium, rubidium, cesium, and francium constitute group one of the periodic tabular array. Although hydrogen is in group 1 (and also in group 17), it is a nonmetal and deserves separate consideration later in this chapter. The name brine metallic is in reference to the fact that these metals and their oxides react with water to form very bones (alkali metal) solutions.
The backdrop of the alkali metals are similar to each other as expected for elements in the aforementioned family unit. The alkali metals take the largest atomic radii and the lowest starting time ionization energy in their periods. This combination makes it very like shooting fish in a barrel to remove the unmarried electron in the outermost (valence) trounce of each. The easy loss of this valence electron means that these metals readily course stable cations with a charge of 1+. Their reactivity increases with increasing atomic number due to the ease of losing the lone valence electron (decreasing ionization energy). Since oxidation is so easy, the contrary, reduction, is difficult, which explains why it is hard to isolate the elements. The solid alkali metals are very soft; lithium, shown in Figure 2, has the lowest density of whatsoever metallic (0.5 g/cm3).
The alkali metals all react vigorously with water to class hydrogen gas and a basic solution of the metal hydroxide. This means they are easier to oxidize than is hydrogen. As an example, the reaction of lithium with water is:
[latex]2\text{Li}(southward)\;+\;2\text{H}_2\text{O}(l)\;{\longrightarrow}\;ii\text{LiOH}(aq)\;+\;\text{H}_2(g)[/latex]

Brine metals react direct with all the nonmetals (except the noble gases) to yield binary ionic compounds containing 1+ metal ions. These metals are and so reactive that it is necessary to avoid contact with both moisture and oxygen in the air. Therefore, they are stored in sealed containers under mineral oil, every bit shown in Figure 3, to prevent contact with air and moisture. The pure metals never be free (uncombined) in nature due to their high reactivity. In addition, this high reactivity makes it necessary to fix the metals by electrolysis of alkali metal compounds.

Different many other metals, the reactivity and softness of the alkali metals make these metals unsuitable for structural applications. However, there are applications where the reactivity of the alkali metals is an advantage. For instance, the production of metals such as titanium and zirconium relies, in part, on the power of sodium to reduce compounds of these metals. The manufacture of many organic compounds, including certain dyes, drugs, and perfumes, utilizes reduction by lithium or sodium.
Sodium and its compounds impart a bright yellow color to a flame, every bit seen in Effigy 4. Passing an electrical discharge through sodium vapor besides produces this color. In both cases, this is an example of an emission spectrum as discussed in the chapter on electronic structure. Streetlights former employ sodium vapor lights because the sodium vapor penetrates fog amend than most other lite. This is because the fog does non besprinkle yellow light equally much equally it scatters white light. The other alkali metals and their salts also impart colour to a flame. Lithium creates a bright, blood-red colour, whereas the others create a stake, violet color.


This video demonstrates the reactions of the alkali metals with h2o.
Grouping 2: The Element of group i Earth Metals
The alkaline world metals (beryllium, magnesium, calcium, strontium, barium, and radium) institute group 2 of the periodic table. The name element of group i metal comes from the fact that the oxides of the heavier members of the grouping react with water to form alkali metal solutions. The nuclear charge increases when going from group one to group 2. Because of this charge increase, the atoms of the alkaline earth metals are smaller and have higher start ionization energies than the alkali metals within the same flow. The college ionization energy makes the alkali metal earth metals less reactive than the alkali metals; however, they are however very reactive elements. Their reactivity increases, as expected, with increasing size and decreasing ionization energy. In chemic reactions, these metals readily lose both valence electrons to form compounds in which they exhibit an oxidation state of 2+. Due to their high reactivity, it is mutual to produce the alkaline earth metals, similar the brine metals, by electrolysis. Even though the ionization energies are low, the ii metals with the highest ionization energies (beryllium and magnesium) do form compounds that exhibit some covalent characters. Like the alkali metals, the heavier alkaline metal earth metals impart color to a flame. Equally in the case of the brine metals, this is role of the emission spectrum of these elements. Calcium and strontium produce shades of crimson, whereas barium produces a greenish color.
Magnesium is a silverish-white metal that is malleable and ductile at high temperatures. Passivation decreases the reactivity of magnesium metallic. Upon exposure to air, a tightly adhering layer of magnesium oxycarbonate forms on the surface of the metallic and inhibits further reaction. (The carbonate comes from the reaction of carbon dioxide in the atmosphere.) Magnesium is the lightest of the widely used structural metals, which is why most magnesium product is for lightweight alloys.
Magnesium (shown in Figure v), calcium, strontium, and barium react with water and air. At room temperature, barium shows the most vigorous reaction. The products of the reaction with h2o are hydrogen and the metal hydroxide. The formation of hydrogen gas indicates that the heavier alkaline earth metals are better reducing agents (more hands oxidized) than is hydrogen. As expected, these metals react with both acids and nonmetals to grade ionic compounds. Unlike virtually salts of the alkali metals, many of the common salts of the alkaline metal earth metals are insoluble in water considering of the high lattice energies of these compounds, containing a divalent metallic ion.

The potent reducing power of hot magnesium is useful in preparing some metals from their oxides. Indeed, magnesium's affinity for oxygen is then great that burning magnesium reacts with carbon dioxide, producing elemental carbon:
[latex]two\text{Mg}(s)\;+\;\text{CO}_2(g)\;{\longrightarrow}\;2\text{MgO}(south)\;+\;\text{C}(s)[/latex]
For this reason, a CO2 fire extinguisher will non extinguish a magnesium fire. Additionally, the brilliant white light emitted by burning magnesium makes it useful in flares and fireworks.
Grouping 12
The elements in group 12 are transition elements; even so, the concluding electron added is non a d electron, just an southward electron. Since the last electron added is an s electron, these elements qualify as representative metals, or post-transition metals. The group 12 elements behave more like the alkali metal earth metals than transition metals. Group 12 contains the 4 elements zinc, cadmium, mercury, and copernicium. Each of these elements has two electrons in its outer vanquish (ns 2). When atoms of these metals course cations with a charge of ii+, where the ii outer electrons are lost, they have pseudo-noble gas electron configurations. Mercury is sometimes an exception because it also exhibits an oxidation state of 1+ in compounds that incorporate a diatomic [latex]\text{Hg}_2^{\;\;two+}[/latex] ion. In their elemental forms and in compounds, cadmium and mercury are both toxic.
Zinc is the most reactive in grouping 12, and mercury is the least reactive. (This is the reverse of the reactivity trend of the metals of groups 1 and ii, in which reactivity increases down a group. The increase in reactivity with increasing atomic number only occurs for the metals in groups 1 and ii.) The decreasing reactivity is due to the formation of ions with a pseudo-noble gas configuration and to other factors that are beyond the telescopic of this give-and-take. The chemic behaviors of zinc and cadmium are quite like to each other but differ from that of mercury.
Zinc and cadmium have lower reduction potentials than hydrogen, and, like the alkali metals and alkaline globe metals, they will produce hydrogen gas when they react with acids. The reaction of zinc with muriatic acid, shown in Effigy vi, is:
[latex]\text{Zn}(s)\;+\;2\text{H}_3\text{O}^+(aq)\;+\;two\text{Cl}^{-}(aq)\;{\longrightarrow}\;\text{H}_2(m)\;+\;\text{Zn}^{ii+}(aq)\;+\;ii\text{Cl}^{-}(aq)\;+\;2\text{H}_2\text{O}(l)[/latex]

Zinc is a silvery metallic that chop-chop tarnishes to a blueish-gray appearance. This change in color is due to an adherent coating of a basic carbonate, Zn2(OH)2CO3, which passivates the metal to inhibit further corrosion. Dry out jail cell and alkaline metal batteries contain a zinc anode. Brass (Cu and Zn) and some bronze (Cu, Sn, and sometimes Zn) are important zinc alloys. Nearly half of zinc production serves to protect fe and other metals from corrosion. This protection may take the grade of a sacrificial anode (as well known as a galvanic anode, which is a means of providing cathodic protection for diverse metals) or every bit a thin blanket on the protected metal. Galvanized steel is steel with a protective coating of zinc.
Sacrificial Anodes
A sacrificial anode, or galvanic anode, is a means of providing cathodic protection of various metals. Cathodic protection refers to the prevention of corrosion by converting the corroding metal into a cathode. As a cathode, the metal resists corrosion, which is an oxidation process. Corrosion occurs at the sacrificial anode instead of at the cathode.
The construction of such a arrangement begins with the attachment of a more active metallic (more negative reduction potential) to the metal needing protection. Zipper may be directly or via a wire. To complete the excursion, a table salt bridge is necessary. This salt bridge is often seawater or ground water. Once the circuit is consummate, oxidation (corrosion) occurs at the anode and not the cathode.
The commonly used sacrificial anodes are magnesium, aluminum, and zinc. Magnesium has the most negative reduction potential of the three and serves best when the common salt bridge is less efficient due to a depression electrolyte concentration such every bit in freshwater. Zinc and aluminum work ameliorate in saltwater than does magnesium. Aluminum is lighter than zinc and has a higher capacity; nevertheless, an oxide coating may passivate the aluminum. In special cases, other materials are useful. For example, atomic number 26 will protect copper.
Mercury is very different from zinc and cadmium. Mercury is the simply metal that is liquid at 25 °C. Many metals dissolve in mercury, forming solutions called amalgams (see the characteristic on Amalgams), which are alloys of mercury with one or more other metals. Mercury, shown in Figure 7, is a nonreactive element that is more difficult to oxidize than hydrogen. Thus, it does not displace hydrogen from acids; however, information technology will react with strong oxidizing acids, such as nitric acrid:
[latex]\text{Hg}(l)\;+\;\text{HCl}(aq)\;{\longrightarrow}\;\text{no\;reaction}[/latex]
[latex]three\text{Hg}(l)\;+\;eight\text{HNO}_3(aq)\;{\longrightarrow}\;3\text{Hg(NO}_3)_2(aq)\;+\;4\text{H}_2\text{O}(l)\;+\;2\text{NO}(g)[/latex]
The clear NO initially formed quickly undergoes further oxidation to the ruby-red dark-brown NO2.

Most mercury compounds decompose when heated. Most mercury compounds contain mercury with a 2+-oxidation state. When there is a large excess of mercury, information technology is possible to grade compounds containing the [latex]\text{Hg}_2^{\;\;two+}[/latex] ion. All mercury compounds are toxic, and information technology is necessary to exercise great care in their synthesis.
Amalgams
An constructing is an blend of mercury with ane or more other metals. This is like to considering steel to be an alloy of iron with other metals. Nearly metals will form an amalgam with mercury, with the main exceptions being fe, platinum, tungsten, and tantalum.
Due to toxicity issues with mercury, there has been a significant decrease in the use of amalgams. Historically, amalgams were important in electrolytic cells and in the extraction of gold. Amalgams of the brine metals still find utilise because they are strong reducing agents and easier to handle than the pure brine metals.
Prospectors had a problem when they plant finely divided gold. They learned that adding mercury to their pans collected the gilded into the mercury to grade an constructing for easier collection. Unfortunately, losses of small amounts of mercury over the years left many streams in California polluted with mercury.
Dentists use amalgams containing argent and other metals to fill cavities. There are several reasons to use an constructing including low cost, ease of manipulation, and longevity compared to alternate materials. Dental amalgams are approximately fifty% mercury by weight, which, in recent years, has become a concern due to the toxicity of mercury.
Later reviewing the best available data, the Food and Drug Assistants (FDA) considers amalgam-based fillings to be rubber for adults and children over six years of age. Even with multiple fillings, the mercury levels in the patients remain far beneath the lowest levels associated with harm. Clinical studies have found no link between dental amalgams and health issues. Health problems may not be the same in cases of children under 6 or pregnant women. The FDA conclusions are in line with the opinions of the Ecology Protection Agency (EPA) and Centers for Disease Command (CDC). The only wellness consideration noted is that some people are allergic to the constructing or ane of its components.
Grouping 13
Group 13 contains the metalloid boron and the metals aluminum, gallium, indium, and thallium. The lightest chemical element, boron, is semiconducting, and its binary compounds tend to be covalent and not ionic. The remaining elements of the group are metals, but their oxides and hydroxides change characters. The oxides and hydroxides of aluminum and gallium exhibit both acidic and basic behaviors. A substance, such equally these 2, that will react with both acids and bases is amphoteric. This characteristic illustrates the combination of nonmetallic and metal behaviors of these two elements. Indium and thallium oxides and hydroxides exhibit merely basic beliefs, in accordance with the clearly metallic grapheme of these 2 elements. The melting bespeak of gallium is unusually depression (about thirty °C) and will melt in your hand.
Aluminum is amphoteric considering information technology will react with both acids and bases. A typical reaction with an acrid is:
[latex]2\text{Al}(s)\;+\;six\text{HCl}(aq)\;{\longrightarrow}\;2\text{AlCl}_3(aq)\;+\;3\text{H}_2(g)[/latex]
The products of the reaction of aluminum with a base of operations depend upon the reaction conditions, with the following being one possibility:
[latex]ii\text{Al}(due south)\;+\;2\text{NaOH}(aq)\;+\;6\text{H}_2\text{O}(l)\;{\longrightarrow}\;2\text{Na[Al(OH)}_4](aq)\;+\;3\text{H}_2(thousand)[/latex]
With both acids and bases, the reaction with aluminum generates hydrogen gas.
The group xiii elements have a valence crush electron configuration of ns 2 np 1. Aluminum commonly uses all of its valence electrons when information technology reacts, giving compounds in which it has an oxidation land of 3+. Although many of these compounds are covalent, others, such as AlF3 and Al2(So4)iii, are ionic. Aqueous solutions of aluminum salts contain the cation [latex]\text{[Al(H}_2\text{O})_6]^{three+}[/latex], abbreviated as Al3+(aq). Gallium, indium, and thallium also course ionic compounds containing Chiliadiii+ ions. These three elements exhibit not only the expected oxidation state of 3+ from the three valence electrons just also an oxidation land (in this case, 1+) that is 2 below the expected value. This phenomenon, the inert pair effect, refers to the germination of a stable ion with an oxidation country two lower than expected for the group. The pair of electrons is the valence s orbital for those elements. In general, the inert pair outcome is important for the lower p-cake elements. In an aqueous solution, the Tl+(aq) ion is more stable than is Tliii+(aq). In full general, these metals will react with air and water to form three+ ions; however, thallium reacts to requite thallium(I) derivatives. The metals of group 13 all react straight with nonmetals such as sulfur, phosphorus, and the halogens, forming binary compounds.
The metals of group 13 (Al, Ga, In, and Tl) are all reactive. However, passivation occurs as a tough, hard, thin pic of the metallic oxide forms upon exposure to air. Disruption of this film may counter the passivation, assuasive the metal to react. 1 manner to disrupt the film is to expose the passivated metallic to mercury. Some of the metal dissolves in the mercury to form an amalgam, which sheds the protective oxide layer to betrayal the metallic to further reaction. The germination of an amalgam allows the metal to react with air and water.

Although hands oxidized, the passivation of aluminum makes information technology very useful as a strong, lightweight building material. Considering of the formation of an amalgam, mercury is corrosive to structural materials made of aluminum. This video demonstrates how the integrity of an aluminum axle can be destroyed past the addition of a small amount of elemental mercury.
The most of import uses of aluminum are in the construction and transportation industries, and in the industry of aluminum cans and aluminum foil. These uses depend on the lightness, toughness, and forcefulness of the metallic, besides as its resistance to corrosion. Considering aluminum is an fantabulous conductor of rut and resists corrosion, it is useful in the industry of cooking utensils.
Aluminum is a very skillful reducing agent and may supercede other reducing agents in the isolation of certain metals from their oxides. Although more expensive than reduction past carbon, aluminum is important in the isolation of Mo, W, and Cr from their oxides.
Grouping 14
The metallic members of group xiv are tin, lead, and flerovium. Carbon is a typical nonmetal. The remaining elements of the group, silicon and germanium, are examples of semimetals or metalloids. Tin can and atomic number 82 grade the stable divalent cations, Sn2+ and Pb2+, with oxidation states ii below the group oxidation country of 4+. The stability of this oxidation country is a consequence of the inert pair upshot. Tin and atomic number 82 also form covalent compounds with a formal iv+-oxidation state. For case, SnCl4 and PbCl4 are low-humid covalent liquids.

Can reacts readily with nonmetals and acids to form tin(Two) compounds (indicating that it is more hands oxidized than hydrogen) and with nonmetals to class either tin(II) or tin(Iv) compounds (shown in Figure viii), depending on the stoichiometry and reaction conditions. Atomic number 82 is less reactive. It is merely slightly easier to oxidize than hydrogen, and oxidation normally requires a hot concentrated acid.
Many of these elements exist every bit allotropes. Allotropes are two or more forms of the same element in the same concrete state with different chemical and concrete properties. In that location are two common allotropes of tin. These allotropes are gray (brittle) tin and white tin. As with other allotropes, the difference between these forms of can is in the arrangement of the atoms. White tin is stable above 13.2 °C and is malleable like other metals. At low temperatures, gray tin is the more stable class. Gray tin is breakable and tends to break down to a powder. Consequently, articles made of tin volition disintegrate in common cold weather, particularly if the cold spell is lengthy. The alter progresses slowly from the spot of origin, and the gray tin can that is first formed catalyzes further change. In a way, this effect is similar to the spread of an infection in a plant or fauna body, leading people to call this process tin affliction or tin pest.
The master utilise of tin is in the coating of steel to form can plate-sheet iron, which constitutes the tin in tin cans. Important can alloys are statuary (Cu and Sn) and solder (Sn and Pb). Lead is important in the lead storage batteries in automobiles.
Group xv
Bismuth, the heaviest member of group fifteen, is a less reactive metal than the other representative metals. It readily gives upwards 3 of its five valence electrons to active nonmetals to grade the tri-positive ion, Biiii+. It forms compounds with the group oxidation state of 5+ merely when treated with strong oxidizing agents. The stability of the 3+-oxidation state is another example of the inert pair consequence.
Cardinal Concepts and Summary
This section focuses on the periodicity of the representative elements. These are the elements where the electrons are entering the s and p orbitals. The representative elements occur in groups 1, ii, and 12–xviii. These elements are representative metals, metalloids, and nonmetals. The alkali metals (grouping i) are very reactive, readily class ions with a accuse of one+ to form ionic compounds that are usually soluble in water, and react vigorously with water to class hydrogen gas and a basic solution of the metal hydroxide. The outermost electrons of the alkaline globe metals (group 2) are more difficult to remove than the outer electron of the alkali metals, leading to the group 2 metals being less reactive than those in group 1. These elements easily course compounds in which the metals exhibit an oxidation state of two+. Zinc, cadmium, and mercury (group 12) unremarkably showroom the group oxidation state of two+ (although mercury as well exhibits an oxidation country of 1+ in compounds that contain [latex]\text{Hg}_2^{\;\;two+}[/latex]). Aluminum, gallium, indium, and thallium (group 13) are easier to oxidize than is hydrogen. Aluminum, gallium, and indium occur with an oxidation country 3+ (however, thallium also commonly occurs equally the Tl+ ion). Tin and lead grade stable divalent cations and covalent compounds in which the metals exhibit the iv+-oxidation state.
Chemical science End of Affiliate Exercises
- How exercise alkali metals differ from alkaline earth metals in atomic structure and full general properties?
- Why does the reactivity of the alkali metals decrease from cesium to lithium?
- Predict the formulas for the nine compounds that may form when each species in column 1 of Table 1 reacts with each species in column 2.
1 2 Na I Sr Se Al O Tabular array 1. - Predict the all-time selection in each of the following. Yous may wish to review the chapter on electronic structure for relevant examples.
(a) the almost metallic of the elements Al, Be, and Ba
(b) the almost covalent of the compounds NaCl, CaCl2, and BeCl2
(c) the lowest get-go ionization energy among the elements Rb, 1000, and Li
(d) the smallest among Al, Al+, and Al3+
(e) the largest amidst Cs+, Ba2+, and Xe
- Sodium chloride and strontium chloride are both white solids. How could you lot distinguish i from the other?
- The reaction of quicklime, CaO, with water produces slaked lime, Ca(OH)2, which is widely used in the construction industry to brand mortar and plaster. The reaction of quicklime and h2o is highly exothermic:
[latex]\text{CaO}(s)\;+\;\text{H}_2\text{O}(50)\;{\longrightarrow}\;\text{Ca(OH)}_2(s)\;\;\;\;\;\;{\Delta}\text{H} = -350\;\text{kJ\;mol}^{-i}[/latex]
(a) What is the enthalpy of reaction per gram of quicklime that reacts?
(b) How much heat, in kilojoules, is associated with the product of 1 ton of slaked lime?
- Write a balanced equation for the reaction of elemental strontium with each of the following:
(a) oxygen
(b) hydrogen bromide
(c) hydrogen
(d) phosphorus
(due east) water
- How many moles of ionic species are nowadays in 1.0 L of a solution marked 1.0 M mercury(I) nitrate?
- What is the mass of fish, in kilograms, that one would take to consume to obtain a fatal dose of mercury, if the fish contains thirty parts per million of mercury by weight? (Assume that all the mercury from the fish ends up equally mercury(Ii) chloride in the trunk and that a fatal dose is 0.20 g of HgClii.) How many pounds of fish is this?
- The elements sodium, aluminum, and chlorine are in the aforementioned period.
(a) Which has the greatest electronegativity?
(b) Which of the atoms is smallest?
(c) Write the Lewis structure for the simplest covalent compound that can course between aluminum and chlorine.
(d) Will the oxide of each element be acidic, basic, or amphoteric?
- Does metallic tin react with HCl?
- What is can pest, also known as tin affliction?
- Compare the nature of the bonds in PbCl2 to that of the bonds in PbClfour.
- Is the reaction of rubidium with water more or less vigorous than that of sodium? How does the rate of reaction of magnesium compare?
Glossary
- alkaline world metal
- whatever of the metals (beryllium, magnesium, calcium, strontium, barium, and radium) occupying grouping 2 of the periodic tabular array; they are reactive, divalent metals that form basic oxides
- allotropes
- two or more forms of the same element, in the same physical state, with different chemical structures
- bismuth
- heaviest fellow member of group 15; a less reactive metal than other representative metals
- metal (representative)
- atoms of the metallic elements of groups 1, 2, 12, 13, 14, 15, and 16, which grade ionic compounds by losing electrons from their outer s or p orbitals
- metalloid
- element that has properties that are between those of metals and nonmetals; these elements are typically semiconductors
- passivation
- metals with a protective nonreactive picture show of oxide or other compound that creates a bulwark for chemical reactions; physical or chemical removal of the passivating motion-picture show allows the metals to demonstrate their expected chemic reactivity
- representative element
- element where the due south and p orbitals are filling
- representative metallic
- metal among the representative elements
Solutions
Answers to Chemical science Finish of Chapter Exercises
1. The alkali metals all have a single s electron in their outermost shell. In dissimilarity, the alkaline metal globe metals have a completed southward subshell in their outermost beat. In full general, the alkali metals react faster and are more reactive than the respective alkaline earth metals in the same period.
three.
[latex]\text{Na}\;+\;\text{I}_2\;{\longrightarrow}\;two\text{NaI} \\[0.5em] 2\text{Na}\;+\;\text{Se}\;{\longrightarrow}\;\text{Na}_2\text{Se} \\[0.5em] 2\text{Na}\;+\;\text{O}_2\;{\longrightarrow}\;\text{Na}_2\text{O}_2 \\[i.5em] \text{Sr}\;+\;\text{I}_2\;{\longrightarrow}\;\text{SrI}_2 \\[0.5em] \text{Sr}\;+\;\text{Se}\;{\longrightarrow}\;\text{SrSe} \\[0.5em] 2\text{Sr}\;+\;\text{O}_2\;{\longrightarrow}\;ii\text{SrO} \\[1.5em] 2\text{Al}\;+\;3\text{I}_2\;{\longrightarrow}\;two\text{AlI}_3 \\[0.5em] 2\text{Al}\;+\;3\text{Se}\;{\longrightarrow}\;\text{Al}_2\text{Se}_3 \\[0.5em] 4\text{Al}\;+\;three\text{O}_2\;{\longrightarrow}\;2\text{Al}_2\text{O}_3[/latex]
5. The possible ways of distinguishing between the ii include infrared spectroscopy past comparison of known compounds, a flame test that gives the characteristic yellow colour for sodium (strontium has a red flame), or comparison of their solubilities in water. At xx °C, NaCl dissolves to the extent of [latex]\frac{35.7\;\text{g}}{100\;\text{mL}}[/latex] compared with [latex]\frac{53.8\;\text{thousand}}{100\;\text{mL}}[/latex] for SrClii. Heating to 100 °C provides an easy test, since the solubility of NaCl is [latex]\frac{39.12\;\text{g}}{100\;\text{mL}}[/latex], but that of SrCl2 is [latex]\frac{100.viii\;\text{g}}{100\;\text{mL}}[/latex]. Density determination on a solid is sometimes difficult, but there is enough departure (2.165 thou/mL NaCl and three.052 g/mL SrCltwo) that this method would exist viable and perchance the easiest and least expensive test to perform.
7. (a) [latex]2\text{Sr}(due south)\;+\;\text{O}_2(grand)\;{\longrightarrow}\;2\text{SrO}(s)[/latex]; (b) [latex]\text{Sr}(south)\;+\;ii\text{HBr}(g)\;{\longrightarrow}\;\text{SrBr}_2(s)\;+\;\text{H}_2(chiliad)[/latex]; (c) [latex]\text{Sr}(southward)\;+\;\text{H}_2(thou)\;{\longrightarrow}\;\text{SrH}_2(s)[/latex]; (d) [latex]6\text{Sr}(s)\;+\;\text{P}_4(s)\;{\longrightarrow}\;2\text{Sr}_3\text{P}_2(s)[/latex]; (e) [latex]\text{Sr}(south)\;+\;2\text{H}_2\text{O}(fifty)\;{\longrightarrow}\;\text{Sr(OH})_2(aq)\;+\;\text{H}_2(g)[/latex]
9. 11 lb
11. Yes, tin reacts with hydrochloric acid to produce hydrogen gas.
13. In PbCl2, the bonding is ionic, as indicated by its melting point of 501 °C. In PbCl4, the bonding is covalent, equally evidenced by it existence an unstable liquid at room temperature.
Source: https://opentextbc.ca/chemistry/chapter/18-1-periodicity/
Post a Comment for "Family of Metals With the Second Highest Reactivity"